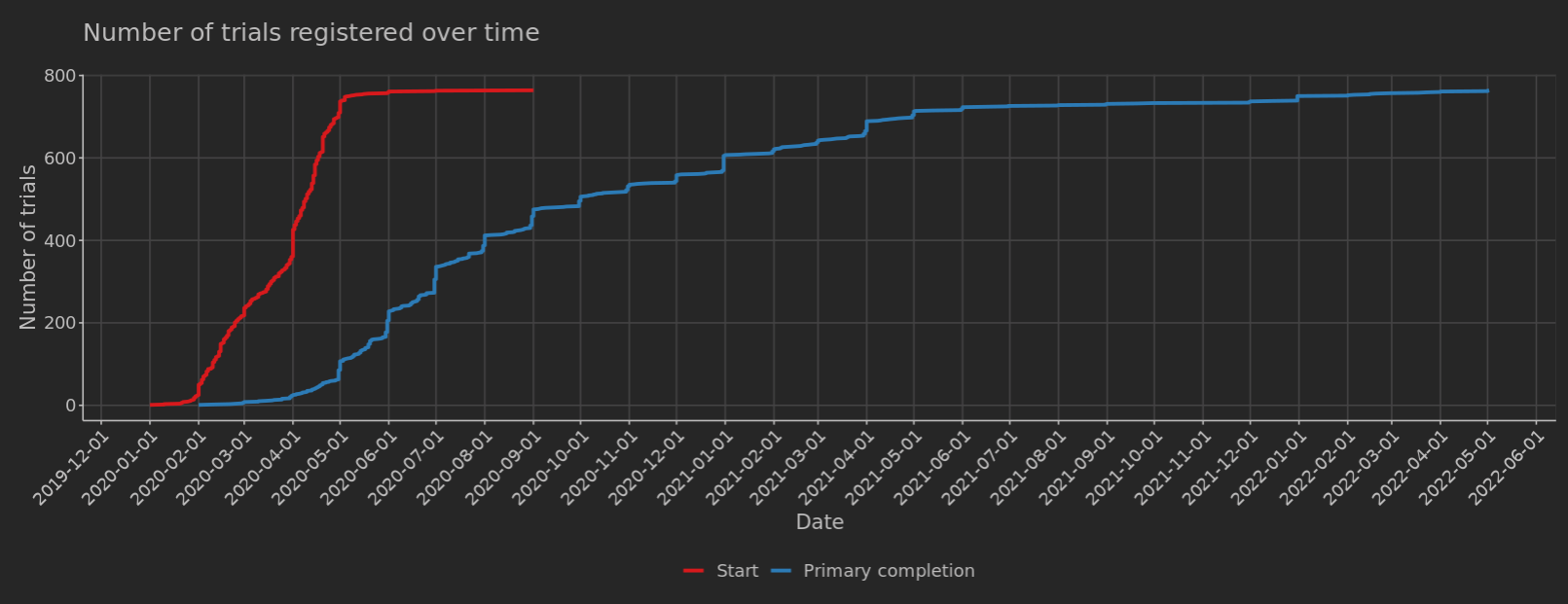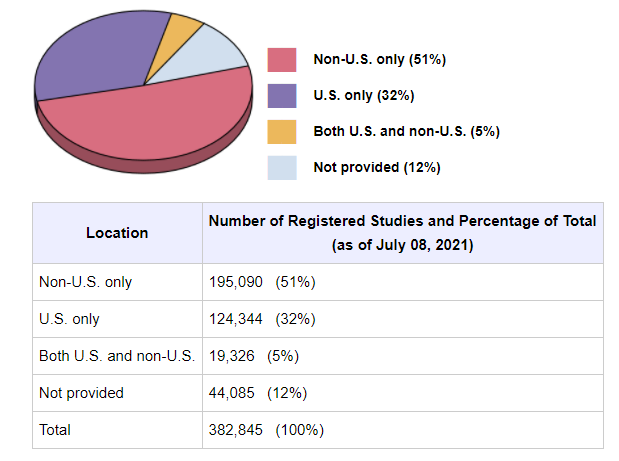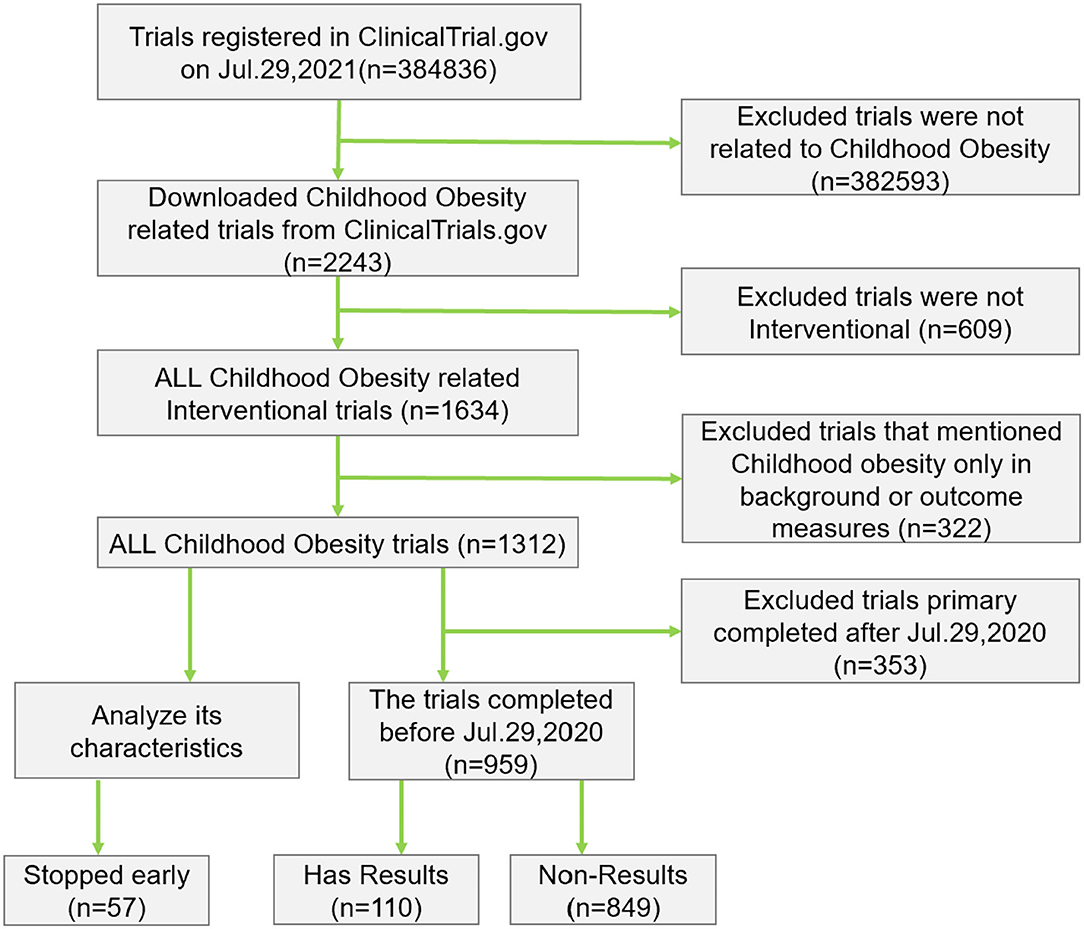
Frontiers | Results Reporting and Early Termination of Childhood Obesity Trials Registered on ClinicalTrials.gov

A group of parents are officially lobbying to register their children as part of a 'control group' for the Covid-19 vaccine. : r/insaneparents
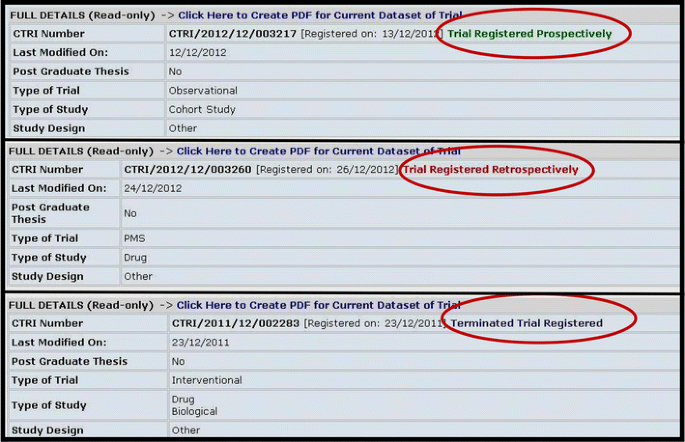
Challenges in Administering a Clinical Trials Registry: Lessons from the Clinical Trials Registry-India | SpringerLink

Registration of Clinical Trials: Background and Implementation Ida Sim, MD, PhD Project Coordinator Department of Research Policy and Cooperation World. - ppt download
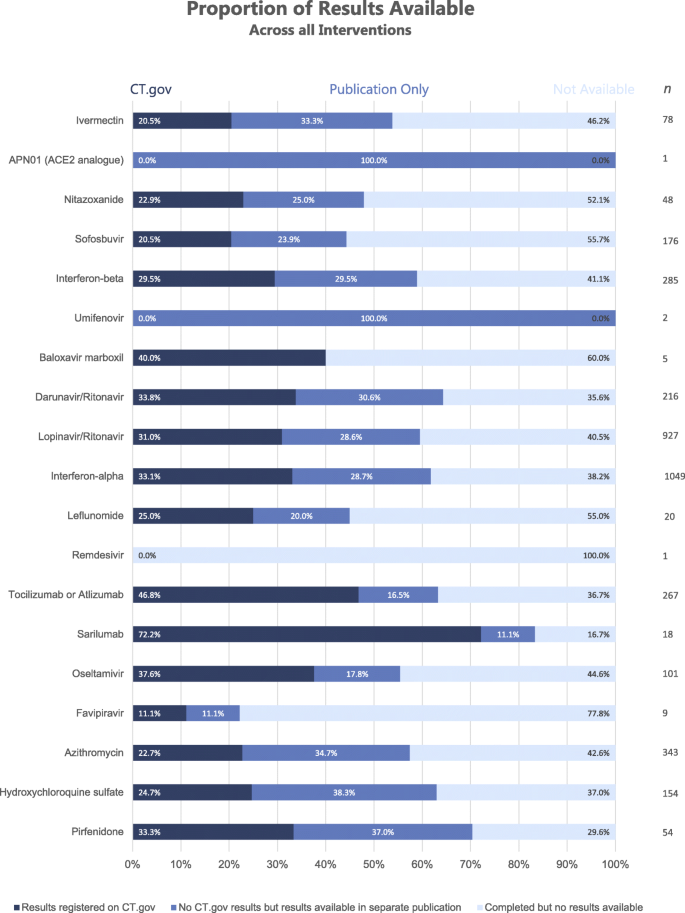
Missing clinical trial data: the evidence gap in primary data for potential COVID-19 drugs | Trials | Full Text

PDF) Using the Cochrane Central Register of Controlled Trials to identify clinical trial registration is insufficient: A cross-sectional study
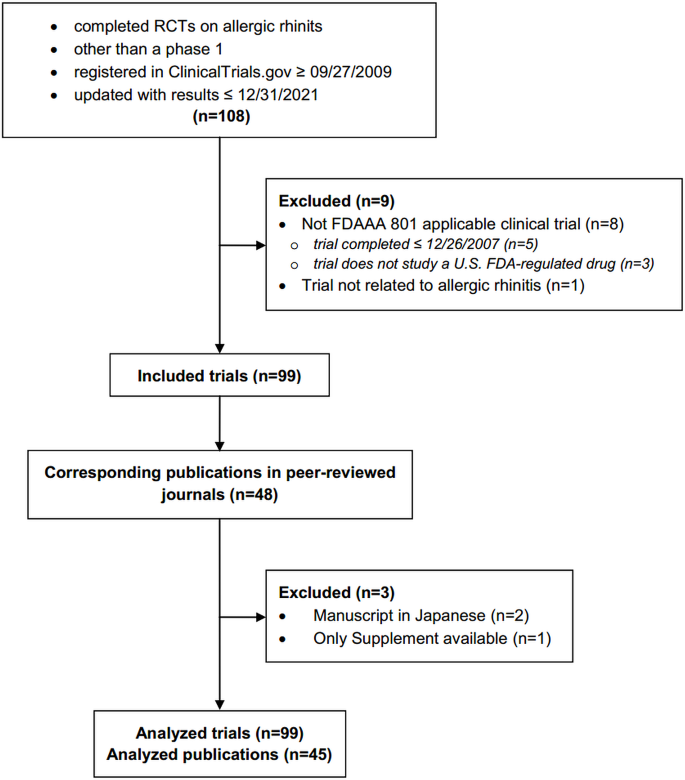
Reporting of the safety from allergic rhinitis trials registered on ClinicalTrials.gov and in publications: An observational study | BMC Medical Research Methodology | Full Text

Regulatory Approval – Clinical Trial Medical Monitoring Plan | Online Clinical Research Courses In India